 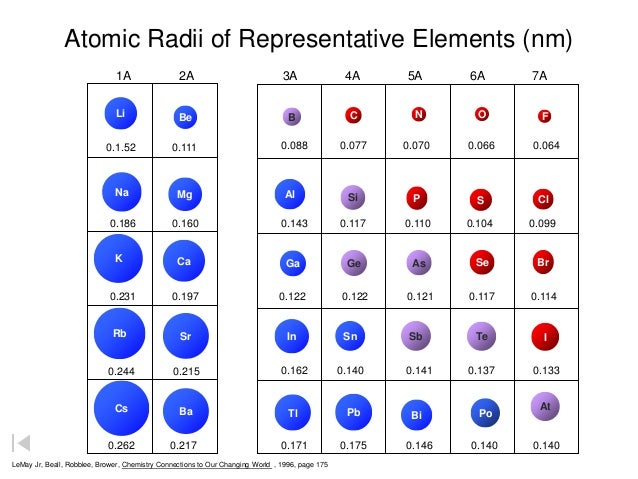
Further information is available in inorganic chemistry textbooks, usually at Level 1 or First Year University level. The terms low spin and high spin refer to the electronic configurations of particular geomtries of certain d-block metal ions. For electronic configurations, where it matters, the values given for octahedral species are low spin unless stated to be high spin. So the correct order of atomic radius is Li < Na < K < Rb < Cs. Li, Na, K, Rb, Cs are IA group elements from Li to Rb the atomic radius will be increased. 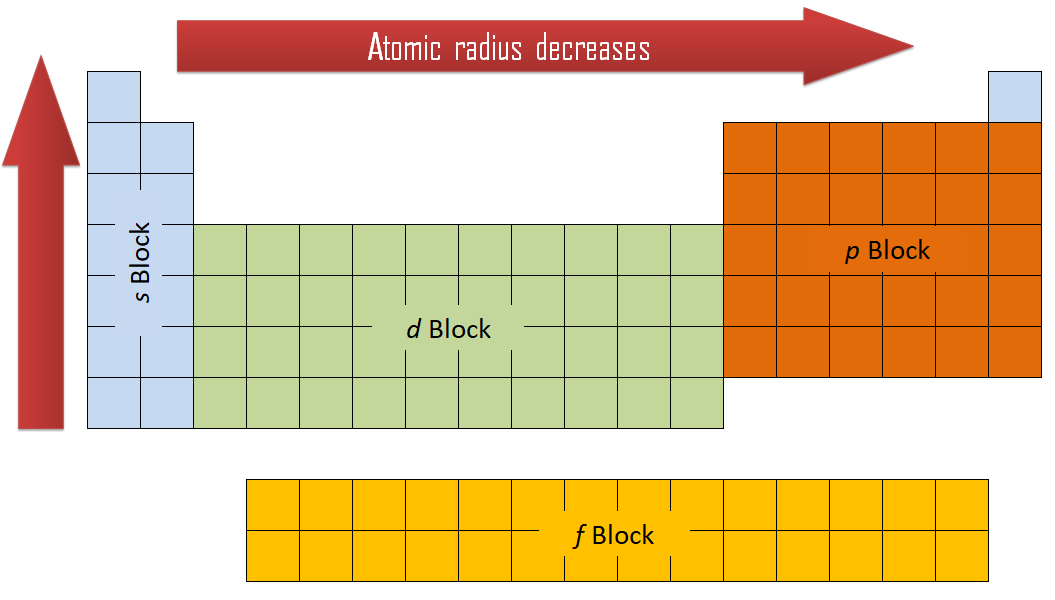
Size does depend upon geometry and environment. Concept: As we move from top to bottom in group the atomic radius will be increased, because the in coming electron enters into a new sub shell. In this table, geometry refers to the arrangment of the ion's nearest neighbours. Hartree-Fock wave functions and radial expectation values: hydrogen to lawrencium, LA-3691, Los Alamos Scientific Laboratory, USA, 1968. 
The R max values for neutral gaseous element valence orbitals are abstracted from reference 1. Image showing periodicity of valence s-orbital radius for the chemical elements as size-coded balls on a periodic table grid. Table: valence shell orbital radii for lithium. Two values are given here, one is based upon calculations and the other upon observation - follow the appropriate link for further details. The problem is its meaning, which is clearly very different in different sources and books. The term "atomic radius" is not particularly helpful although its use is widespread. Follow the appropriate hyperlinks for definitions of each radius type. The size of neutral atoms depends upon the way in which the measurement is made and the environment. All values of radii are given in picometres (pm). Follow the appropriate hyperlinks for literature references and definitions of each type of radius. There are several other ways ways to define radius for atoms and ions. It is not always easy to make sensible comparisons between the elements however as some bonds are quite short because of multiple bonding (for instance the O=O distance in O 2 is short because of the the double bond connecting the two atoms. Note: If we look at the atomic radius trend in a group then it increases when moving from top to down as there is addition of shells in the atom when you move down the group.One measure of size is the element-element distance within the element. Hence, the correct order would be Li > Be > B. The elements Lithium belongs to the first group and Be belongs to the second group but Boron belongs to the 13th group and since they lie in the same 1st period therefore, moving from left to right the atomic radius will decrease. This higher effective nuclear charge pulls the electrons towards itself, which causes the decrease in the radius and hence the lesser atomic radius. When you are moving from left to right in a period then, effective nuclear charge ( the net positive charge, the electrons can feel as the core electrons will cloud the valence electrons to feel the attraction from) increases. Atomic Radius is the distance from the atom’s nucleus to the outermost shell of the atom. Hence, the atomic radius decreases moving from left to right in a period. Hint: >The periodic table is designed in such a way that elements are put in increasing order of their atomic masses and surprisingly, their physical and chemical properties are the same within a group or block. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
